SCOPE 2022 – THE IMPORTANCE OF A CONNECTED ECOSYSTEM?
[Author:
AMY APOSTOLERIS | Global Head, Digital Clinical Trials Solutions]
I hope you all enjoyed SCOPE this year as much as I did! With the many sessions scheduled throughout the four days, both virtual and in person, as well as the exhibitors, I found the conference to be quite dynamic. From live presentations and interactive panels, we heard case studies by industry leading experts, and strategies for digitizing clinical trials post pandemic. There were also key learnings derived across, and especially on the topic of decentralized and digital solutions. From the numerous discussions with biopharmaceutical companies, site networks, CROs, consultants, as well as decentralized clinical trial (DCT) support services and digital product vendors, there is no doubt that our industry is surely planning for the “new normal” — where utilizing digital tools and platforms to conduct clinical trials is no longer a concept, but a reality.
PATIENT ENGAGEMENT, PATIENT CENTRICITY and DATA INSIGHTS were a few of the themes at SCOPE2022 that resonated with me, both during the speaking and panel sessions and in the exhibit hall. The importance of connecting patients to clinical trials “where they are” and collecting patient data remotely and engaging them through digital technology and AI/ML has been a key topic of importance for me and an essential strategy resulting from the Covid pandemic. While determined to find solutions to prevent possible study closings, and the challenges to relaunch, most clinical development and operations teams realized that clinical trials could in fact continue for patients and sites, in ways that are less burdensome for patients, using digital applications, connected devices and SaaS platforms. We also realized a benefit in that digital technology can ultimately make clinical trials more efficient, bring better data quality and even accelerate timelines.
WHY A CONNECTED ECOSYSTEM IS KEY TO PATIENT CENTRICITY?
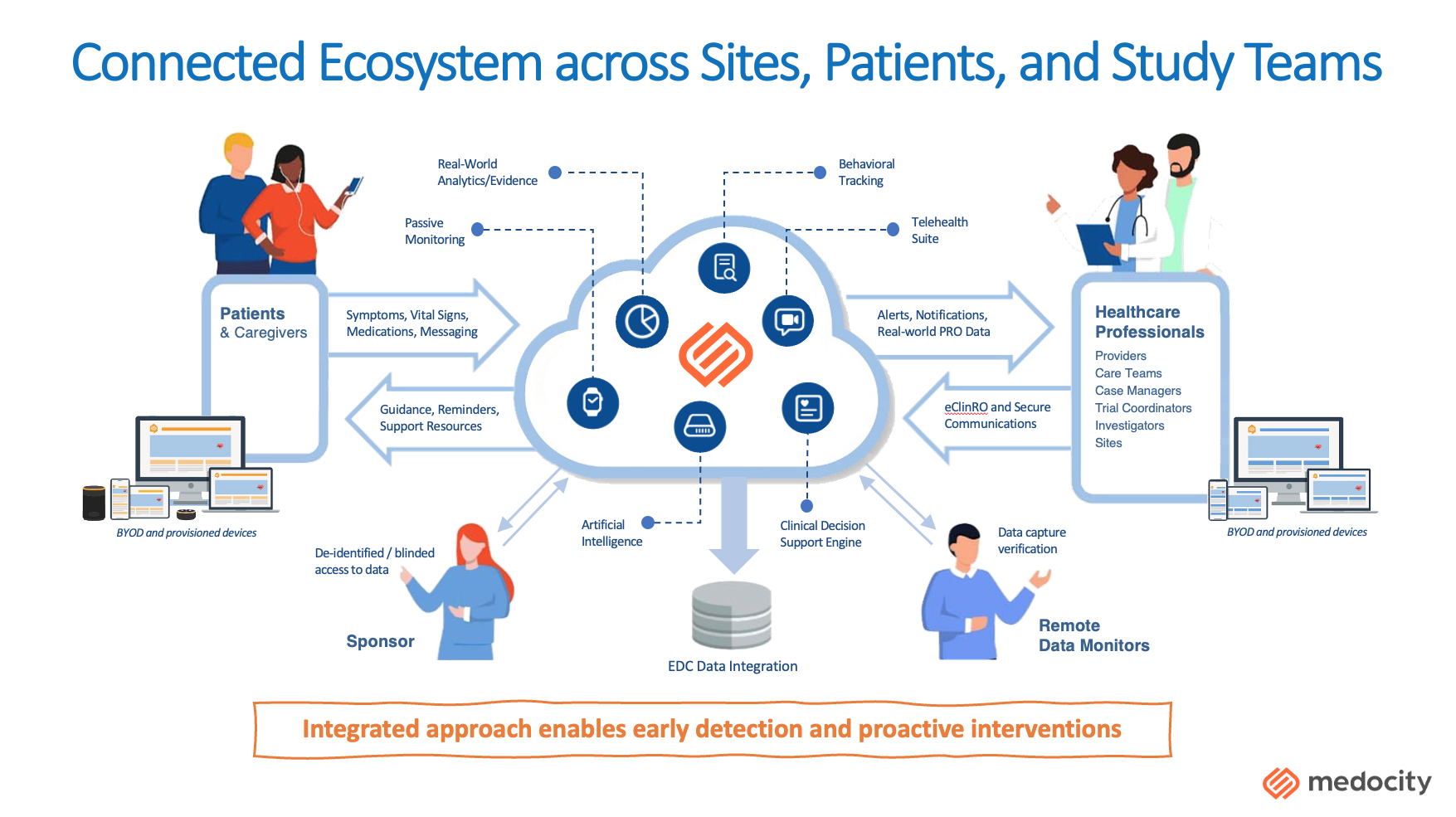
A myriad of service and product vendors attended the conference, from EDC, CTMS, IRT and SaaS platforms and AI/ML solutions. While many are offering single solutions to virtually connect, collect, and track patient data, an unmet need exists for a fully comprehensive, patient-centric, and unified solution that connects all participants and sponsors in the clinical trial ecosystem. A solution that brings connectivity between patients, investigator sites, study teams and sponsors together in real time to enable actionable data monitoring and empower swift decision making.
In a clinical trial connected ecosystem the patient is the focus of the network and empowers the patient to be more engaged and connected to the study protocol as well as their own health in a unified center of clinical research and health care, while providing real-time data and insights that benefit all stakeholders in the trial. From a technology perspective, a clinical trial ecosystem can be defined as a fully interoperable and composable digital SaaS platform that can:
- Ingest patient data and integrate data insights from external data sources
- Support patients throughout their clinical study journey to provide guidance and engagement
- Enable real-time communication and data sharing between patients and investigators sites
- Provide sponsors with study progress visibility and actionable study analytics to empower swift decision making
The benefits of a connected patient ecosystem, as validated through health outcomes research and the Basch study (ASCO, 2017), reveal that when patients and sites are actively engaged and patients are informed through data transparency, study outcomes can improve significantly. Basch’s ground-breaking study demonstrated the importance and impact of patients staying connected as part of their disease and study journey. The results were compelling and game changing, establishing that patients were more compliant, stayed on medication longer and adhered to the study protocol requirements when digital engaged.
Similar findings in terms of patient engagement, compliance, and benefits to study outcomes have been proven with Medocity’s digital platform across chronic and complex diseases. The data continues to demonstrate that the more connected patients are to their health, the study sites and to their progress, the more the patients benefit and the better the study outcomes. Through the utilization of a fully patient centered digital engagement platform, the reality of a connected ecosystem that benefits all stakeholders can be accomplished. Medocity’s AI/ML driven, highly composable and interoperable platform is a game changer in clinical development. Putting the patient at the center, with clinical trial navigation at the palm of their hands, reduces patient burden, accelerates enrollment, and improves adherence and retention.
As we gather these insights from SCOPE and enter this new era of decentralized trials, digital technology, and patient centricity I look forward to continuing the discussion on the importance of focusing on the connected study ecosystem to benefit the patient and study outcomes. And to share with you how the next generation of platforms clearly puts the patient at the center and imposes data quality and integrity for stakeholders. Ultimately our goal is to ingest and share important insights that will guide strategies for future research and development and help bring therapies to patients faster and with lower cost.
Medocity shares our customers’ commitment to patient-centricity as a core value
Medocity was founded to address the needs of oncology patients who are disconnected from care the moment they are discharged from a clinical setting. The Medocity platform has been designed in conjunction with patients to deliver a highly engaging experience that enables real-time connectivity and monitoring for any chronic or complex disease across the care continuum.
Uniquely differentiated capabilities that make a difference in patient’s lives and well-being
Medocity’s clinical rules engine can assess incoming ePRO’s and triage them in real time based on the underlying protocol. Patients who report symptoms, side effects or vital signs get immediate, personalized feedback. Simultaneously, the trial site can set alerts at the population or patient level and then respond in real time using a fully integrated telehealth suite. Nothing is more patient-centric than the potential to drive meaningful interventions the moment a patient appears to be in distress.
In an increasingly digital age, no one platform, even Medocity’s, can address every theoretical aspect of how a trial might be configured to optimize outcomes. That’s why it’s critically important to select a vendor like Medocity with an open architecture and the proven ability to integrate with any device, sensor, wearable, or even platform such as EDC systems, EMR, lab, or specialty pharmacy, to future-proof the underlying technology solution.